|
Suppose we have 100.0 g of 3H (tritium, a radioactive isotope of hydrogen). The half-life of a specific radioactive isotope is constant it is unaffected by conditions and is independent of the initial amount of that isotope.Ĭonsider the following example. 
The half-life of a radioactive isotope is the amount of time it takes for one-half of the radioactive isotope to decay.  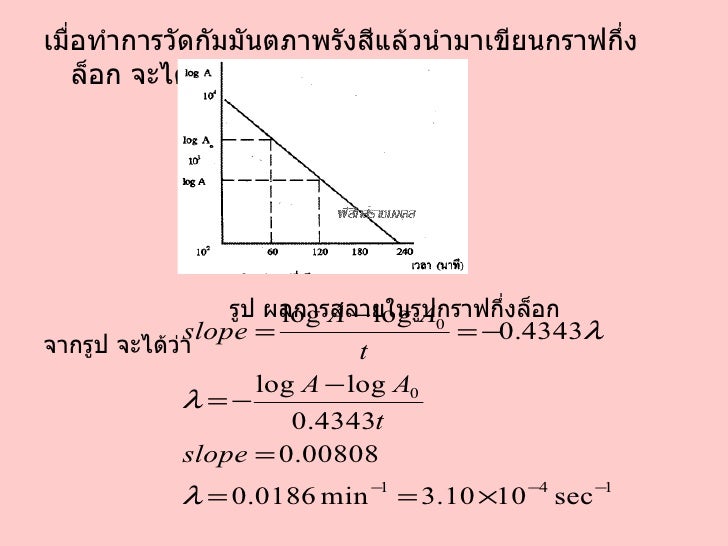
An interesting and useful aspect of radioactive decay is the half-life. As time passes, less and less of the radioactive isotope will be present, and the level of radioactivity decreases. Some isotopes are stable indefinitely, while others are radioactive and decay through a characteristic form of emission. .jpg)
Whether or not a given isotope is radioactive is a characteristic of that particular isotope.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
